Abstract
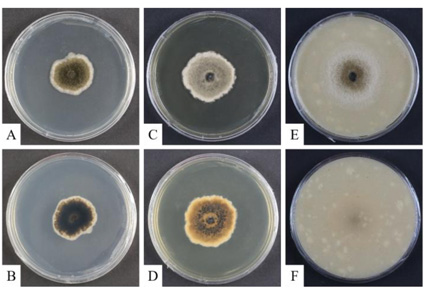
Fungal strains, designated KNU-NL4 and KNU-OL2, belonging to the family Didymellaceae were isolated from a soil sample collected in Miryang, Korea. Phylogenetic analyses based on a concatenated dataset of DNA sequences of ITS regions and partial sequences of ACT, CAL, TEF1-α, and β-TUB genes showed that the isolates reside in a clade together with Boeremia species but occupy the distinct phylogenetic position. Morphologically, the novel strains produce bigger conidiomata (average size 169.8 μm) than the closely related B. rhapontica (126.59 μm) and smaller than the other close neighbor B. coffeae (187.5 μm). Both novel strains also differed from them by smaller colony size and colony color on OA and MEA. The detailed descriptions, illustrations, and discussions regarding the morphological and phylogenetic analyses of the closely related species are provided to support the novelty of the isolated species. The results of phylogenetic analysis and morphological observations indicate that strains KNU-NL4 and KNU-OL2 represent a novel species in the genus Boeremia, for which the name Boeremia parva sp. nov. is proposed.
References
<p>Aveskamp, M.M., Woudenberg, J.H.C., de Gruyter, J., Turco, E., Groenewald, J.Z. & Crous, P.W. (2009) Development of taxon-specific sequence characterized amplified region (SCAR) markers based on actin sequences and DNA amplification fingerprinting (DAF): a case study in the <em>Phoma exigua </em>species complex. <em>Molecular Plant Pathology </em>10 (3): 403–414. https://doi.org/10.1111/J.1364-3703.2009.00540.X</p>
<p>Aveskamp, M.M., de Gruyter, J., Woudenberg, J.H.C., Verkley, G.J.M. & Crous, P.W. (2010) Highlights of the Didymellaceae: A polyphasic approach to characterise <em>Phoma</em> and related pleosporalean genera. <em>Studies in Mycology</em> 65: 1–60. https://doi.org/10.3114/sim.2010.65.01</p>
<p>Berner, D., Cavin, C., Woudenberg, J.H.C., Tunali, B., Büyük, O. & Kansu, B. (2015) Assessment of <em>Boeremia exigua</em> var. <em>rhapontica</em>, as a biological control agent of Russian knapweed (<em>Rhaponticum repens</em>). <em>Biological Control</em> 81: 65–75. https://doi.org/10.1016/j.biocontrol.2014.11.009</p>
<p>Betina, V. (1992) Biological effects of the antibiotic brefeldin A (decumbin, cyanein, ascotoxin, synergisidin): a retrospective. <em>Folia Microbiolica</em> 37 (1): 3–11. https://doi.org/10.1007/BF02814572</p>
<p>Boerema, G.H. (1997) Contributions towards a monograph of <em>Phoma</em> (Coelomycetes) – V. Subdivision of the genus in sections. <em>Mycotaxon</em> 64: 321–333.</p>
<p>Boerema, G.H., de Gruyter, J., Noordeloos, M.E. & Hamers, M.E.C. (2004) <em>Phoma </em>identification manual. Differentiation of specific and infra-specific taxa in culture. CABI Publishing, Wallingford, 470 pp.</p>
<p>Bottalico, A., Capasso, R., Evidente, A. & Vurro, M. (1994) Process for the production and purification of cytochalasin B from <em>Phoma exigua </em>var. <em>heteromorpha</em>. <em>Applied Biochemistry and Biotechnology </em>48 (1): 33–36. https://doi.org/10.1007/BF02825357</p>
<p>Carbone, I. & Kohn, L.M. (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. <em>Mycologia</em> 91: 553–556. https://doi.org/10.1080/00275514.1999.12061051</p>
<p>Cimmino, A., Andolfi, A., Berestetskiy, A. & Evidente, A. (2008) Production of phytotoxins by <em>Phoma exigua</em> var. <em>exigua</em>, a potential mycoherbicide against perennial thistles. <em>Journal of Agriculture Food Chemistry</em> 56 (15): 6304–6309. https://doi.org/10.1021/jf8004178</p>
<p>Che, Y., Gloer, J.B. & Wicklow, D.T. (2002) Phomadecalins A–D and phomapentenone A: new bioactive metabolites from <em>Phoma </em>sp. NRRL 25697, a fungal colonist of <em>Hypoxylon stromata</em>. <em>Journal of Natural Products</em> 65 (3): 399–402. https://doi.org/10.1021/np010519o</p>
<p>Chen, Q., Zhang, K., Zhang, G.Z. & Cai, L. (2015) A polyphasic approach to characterize two novel species of <em>Phoma</em> (<em>Didymellaceae</em>) from China. <em>Phytotaxa</em> 197 (4): 267–281. https://doi.org/10.11646/phytotaxa.197.4.4</p>
<p>Chen, Q., Hou, L.W., Duan, W.J. Crous, P.W. & Cai, L. (2017) Didymellaceae revised. <em>Studies in Mycology</em> 87: 105–156. http://dx.doi.org/10.1016/j.simyco.2017.06.002</p>
<p>Damm, U., Cannon, P.F., Woudenberg, J.H.C., Johnston, P.R., Weir, B.S., Tan, Y.P., Shivas, R.G. & Crous, P.W. (2012) The <em>Colletotrichum boninense</em> species complex. <em>Studies in Mycology</em> 73 (1): 1–36. http://dx.doi.org/10.3114/sim0002</p>
<p>de Gruyter, J. & Scheer, P. (1998) Taxonomy and pathogenicity of <em>Phoma exigua </em>var. <em>populi </em>var<em>. </em>nov. causing necrotic bark lesions on poplars. <em>Journal of Phytopathology</em> 146 (8–9): 411–415. https://doi.org/10.1111/j.1439-0434.1998.tb04773.x</p>
<p>de Gruyter, J., Boerema, G.H. && Van Der Aa, H.A. (2002) Contributions towards a monograph of <em>Phoma</em> (Coelomycetes) VI–2. Section of Phyllostictoides: Outline of its taxa. <em>Persoonia – Molecular Phylogeny and Evolution of Fungi </em>18 (1): 1–53.</p>
<p>de Gruyter, J., Aveskamp, M.M., Woudenberg, J.H.C., Verkley, G.J.M., Groenewald, J.Z. & Crous PW. (2009) Molecular phylogeny of <em>Phoma</em> and allied anamorph genera: Towards a reclassification of the <em>Phoma </em>complex. <em>Mycological Research</em> 113: 508–519. https://doi.org/10.1016/j.mycres.2009.01.002</p>
<p>Felsenstein, J. (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. <em>Molecular Biology and Evolution</em> 17: 368–376. https://doi.org/10.1007/BF01734359</p>
<p>Fitch, W.M. (1971) Toward defining the course of evolution: minimum change for a specific tree topology. <em>Systematic Zoology</em> 20 (4): 406–416. https://doi.org/10.2307/2412116</p>
<p>Glass, N.L. & Donaldson, G.C. (1995) Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. <em>Applied and Environmental Microbiology</em> 61 (4): 1323–1330. http://dx.doi.org/10.1128/aem.61.4.1323-1330.1995</p>
<p>Gorny, A.M., Kikkert, J.R., Dunn, A.R., Dillard, H.R., Smart, C.D. & Pethybridge, S.J. (2015) Tan spot of lima bean caused by <em>Boeremia exigua</em> var. <em>exigua</em> in New York State, USA.<em> Canadian Journal of Plant Pathology</em> 37 (4): 523–528. https://doi.org/10.1080/07060661.2015.1105873</p>
<p>Jayasiri, S.C., Hyde, K.D., Jones, E.B., Jeewon, R., Ariyawansa, H.A., Bhat, J.D., Camporesi, E. & Kang, J.C. (2017) Taxonomy and multigene phylogenetic evaluation of novel species in <em>Boeremia</em> and <em>Epicoccum </em>with new records of <em>Ascochyta </em>and <em>Didymella</em> (<em>Didymellaceae</em>). <em>Mycosphere </em>8 (8): 1080–1101. https://doi.org/10.5943/mycosphere/8/8/9</p>
<p>Jayawardena, R.S., Hyde, K.D., Jeewon, R., Ghobad-Nejhad, M., Wanasinghe, D.N., Liu, N., Phillips, A.J.L., Oliveira-Filho, J.R.C., da Silva, G.A. & Gibertoni, T.B. (2019) One stop shop II: taxonomic update with molecular phylogeny for important phytopathogenic genera: 26-50 (2019). <em>Fungal Diversity</em> 94: 41–129. https://doi.org/10.1007/s13225-019-00418-5</p>
<p>Kimura, M. (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. <em>Molecular Biology and Evolution</em> 16 (2): 111–120. https://doi.org/10.1007/bf01731581</p>
<p>Koike, S.T., Subbarao, K.V., Verkley, G.J., Fogle, D. & OʹNeil, T.M. (2006) <em>Phoma</em> basal rot of Romaine lettuce in California caused by <em>Phoma exigua</em>: Occurrence, characterization, and control. <em>Plant Disease</em> 90 (10): 1268–1275. https://doi.org/10.1094/PD-90-1268</p>
<p>Kowalski, T., Kraj, W., Bednarz, B. & Rossa, R. (2019) The association of <em>Boeremia lilacis</em> with necrotic lesions on shoots and leaf petioles and its pathogenicity towards <em>Fraxinus excelsior</em>. <em>European Journal of Plant Pathology</em> 154 (4): 961–974. https://doi.org/10.1007/s10658-019-01715-0</p>
<p>Kumar, S., Stecher, G. & Tamura, K. (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. <em>Molecular Biology and Evolution</em> 33 (7): 1870–1874. https://doi.org/10.1093/molbev/msw054</p>
<p>Larkin, M.A., Blackshields, G., Brown, N.P., Chenna, R., McGettigan, P.A., McWilliam, H., Valentin, F., Wallace, I.M., Wilm, A., Lopez, R., Thompson, J.D., Gibson, T.J. & Higgins, D.G. (2007) Clustal W and Clustal X version 2.0. <em>Bioinformatics</em> 23 (21): 2947–2948. https://doi.org/10.1093/bioinformatics/btm404</p>
<p>Marin-Felix, Y., Groenewald, J.Z., Cai, L. Chen, Q., Marincowitz, S., Barnes, I., Bensch, K., Braun, U., Camporesi, E., Damm, U., de Beer, Z.W., Dissanayake, A., Edwards, J., Giraldo, A., Hernández-Restrepo, M., Hyde, K.D., Jayawardena, R.S., Lombard, L., Luangsa-ard, J., McTaggart, A.R., Rossman, A.Y., Sandoval-Denis, M., Shen, M., Shivas, R.G., Tan, Y.P., van der Linde, E.J., Wingfield, M.J., Wood, A.R., Zhang, J.Q., Zhang, Y. & Crous, P.W. (2017) Genera of phytopathogenic fungi: GOPHY 1. <em>Studies in Mycology</em> 86: 99–216. https://doi.org/10.1016/j.simyco.2017.04.002</p>
<p>Monte, E., Bridge, P.D. & Sutton, B.C. (1991) An integrated approach to <em>Phoma</em> systematics. <em>Mycopathologia</em> 115 (2): 89–103. https://doi.org/10.1007/BF00436797</p>
<p>O’Donnell, K. & Cigelnik, E. (1997) Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus <em>Fusarium </em>are nonorthologous. <em>Molecular Phylogenetics and Evolution </em>7 (1): 103–116. https://doi.org/10.1006/mpev.1996.0376</p>
<p>O’Donnell, K., Kistler, H.C., Cigelnik, E. & Ploetz, R.C. (1998) Multiple evolutionary origins of the fungus causing Panama disease of banana: concordant evidence from nuclear and mitochondrial gene genealogies. <em>Proceedings of the National Academy of Sciences of the United States of America</em> 95 (5): 2044–2049. https://doi.org/10.1073/pnas.95.5.2044</p>
<p>Park, S., Ten, L., Lee, S.Y., Back, C.G., Lee, J.J., Lee, H.B. & Jung, H.Y. (2017) New recorded species in three genera of the Sordariomycetes in Korea. <em>Mycobiology</em> 45 (2): 64–72. https://doi.org/10.5941/MYCO.2017.45.2.64</p>
<p>Rai, M., Deshmukh, P., Gade, A., Ingle, A., Gyorgy, J. Kovics, G.J. & Irinyi, L. (2009) <em>Phoma Saccardo</em>: distribution, secondary metabolite production and biotechnological applications. <em>Critical Reviews in Microbiology</em> 35 (3): 182–196. https://doi.org/10.1080/10408410902975992</p>
<p>Rai, M., Gade, A., Zimowska, B., Ingle, A.P. & Ingle, P. (2018) Marine-derived phoma-the gold mine of bioactive compounds. <em>Applied Microbiology and Biotechnology</em> 102 (21): 9053–9066. https://doi.org/10.1007/s00253-018-9329-2</p>
<p>Saitou, N. & Nei, M. (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. <em>Molecular Biology and Evolution</em> 4 (4): 406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454</p>
<p>Seifert, K.A. (2009) Progress towards DNA barcoding of fungi. <em>Molecular Ecology Resources</em> 9 (Suppl. 1): 83–89. https://doi.org/10.1111/j.1755-0998.2009.02635.x</p>
<p>Singh, S.B., Zink, D.L., Goetz, M.A., Dombrowski, A.W., Polishook, J.D. & Hazuda, D.J. (1998) Equisetin and a novel opposite stereochemical homolog Phomasetin, two fungal metabolites as inhibitors of HIV-1 integrase. <em>Tetrahedron </em>39 (16): 2243–2246. https://doi.org/10.1016/S0040-4039(98)00269-X</p>
<p>Stewart, R.B. (1957) Leaf blight and stem dieback of coffee caused by an undescribed species of Ascochyta. <em>Mycologia</em> 49 (3): 430–433. https://doi.org/10.1080/00275514.1957.12024659</p>
<p>Wang, L.W., Xu, B.G., Wang, J.Y., Su, Z.Z., Lin, F.C., Zhang, C.L. & Kubicek, C.P. (2012) Bioactive metabolites from <em>Phoma</em> species, an endophytic fungus from the Chinese medicinal plant <em>Arisaema erubescens</em>. <em>Applied Microbiology and Biotechnology</em> 93 (3): 1231–1239. https://doi.org/10.1007/s00253-011-3472-3</p>
<p>White, T.J., Bruns, T., Lee, S. & Taylor, J. (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. <em>In:</em> Innis, M.A., Gelfand, D.H., Sninsky, J.J. & White, T.J. (Eds.) <em>PCR protocols: a guide to methods and applications.</em> Academic Press, San Diego (CA), pp. 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1</p>