Abstract
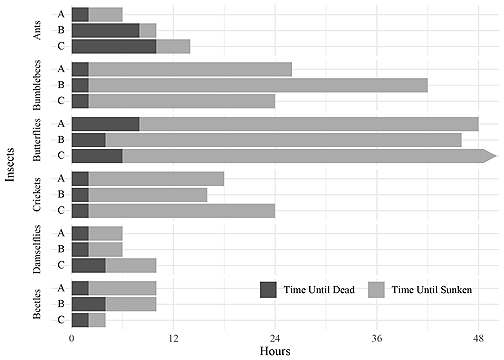
The fossil record represents a small fraction of what lived in ancient ecosystems. Transport, the carrying of organisms from their living environment to their burial environment, remains an enigmatic part of the fossilization process. We analyzed the effects of transport on the preservation potential of a wide variety of insects with different morphologies: Camponotus spp. (carpenter ants), Bombus spp. (bumblebees), Polyommatus spp. (butterflies), Teleogryllus oceanicus (oceanic house crickets), Enallagma spp. (blue damselflies) and Coccinellidae (ladybird beetles). Live specimens were placed in rotary tumblers with water and silt-sized silica sediment for 48 hours. Specimen vitality, buoyancy, and articulation were recorded every two hours to determine the relative preservation potential of these insects. We find that ants, bumblebees, crickets, and ladybird beetles are less likely to disarticulate to unidentifiable degrees—even under long transport times. In contrast, butterflies and damselflies disarticulate relatively rapidly. These results suggest that the more robust group of insects could be overrepresented in environments that would contain many transported specimens, such as fluvial, deltaic, and coastal marine.
References
- Allison, P.A. (1986) Soft-bodied animals in the fossil record: The role of decay in fragmentation during transport. Geology, 14 (12), 979–981. https://doi.org//10.1130/0091-7613(1986)14<979:SAITFR>2.0.CO;2
- Allison, P.A. (1988) The role of anoxia in the decay and mineralization of proteinaceous macro-fossils. Paleobiology, 2 (2), 139–154. https://doi.org//10.1017/S009483730001188X
- Álvarez‐Parra, S., Peñalver, E., Nel, A. & Delclòs, X. (2022) New barklice (Psocodea, Trogiomorpha) from Lower Cretaceous Spanish amber. Papers in Palaeontology, 8 (3), e1436. https://doi.org//10.1002/spp2.1436
- Anderson, E.P. & Smith, D.M. (2017) The same picture through different lenses: Quantifying the effects of two preservation pathways on Green River Formation insects. Paleobiology, 43 (2), 224–247. https://doi.org//10.1017/pab.2016.29
- Archibald, S.B., Bossert, W.H., Greenwood, D.R. & Farrell, B.D. (2010) Seasonality, the latitudinal gradient of diversity, and Eocene insects. Paleobiology, 36 (3), 374–398. https://doi.org//10.1666/09021.1
- Archibald, S.B., Johnson, K.R., Mathewes, R.W. & Greenwood, D.R. (2011) Intercontinental dispersal of giant thermophilic ants across the Arctic during early Eocene hyperthermals. Proceedings of the Royal Society B: Biological Sciences, 278 (1725), 3679–3686.
- https://doi.org/10.1098/rspb.2011.0729
- Archibald, S.B., Morse, G.E., Greenwood, D.R. & Mathewes, R.W. (2014) Fossil palm beetles refine upland winter temperatures in the Early Eocene Climatic Optimum. Proceedings of the National Academy of Sciences of the United States of America, 111 (22), 8095–8100.
- https://doi.org/10.1073/pnas.1323269111
- Archibald, S.B., Ware, J.L., Rasmussen, J.A., Sylvestersen, R.L., Olsen, K. & Simonsen, T.J. (2023) The damselfly genus Furagrion Petrulevičius et al. (Odonata, Zygoptera) from the early Eocene Fur Formation of Denmark and the dysagrionoid grade. Zootaxa, 5278 (2), 289–317. https://doi.org//10.11646/zootaxa.5278.2.4
- Bath Enright, O.G., Minter, N.J. & Sumner, E.J. (2017) Palaeoecological implications of the preservation potential of soft-bodied organisms in sediment-density flows: testing turbulent waters. Royal Society Open Science, 4 (6), 170212. https://doi.org//10.1098/rsos.170212
- Bath Enright, O.G., Minter, N.J., Sumner, E.J., Mángano, M.G. & Buatois, L.A. (2021) Flume experiments reveal flows in the Burgess Shale can sample and transport organisms across substantial distances. Communications Earth & Environment, 2 (1), 104. https://doi.org//10.1038/s43247-021-00176-w
- Behrensmeyer, A.K., Kidwell, S.M. & Gastaldo, R.A. (2000) Taphonomy and paleobiology. Paleobiology, 26 (S4), 103–147. https://doi.org//10.1017/S0094837300026907
- Bello, E., Chen, Y. & Alleyne, M. (2023) Staying dry and clean: An insect’s guide to hydrophobicity. Insects, 14 (1), 42. https://doi.org//10.3390/insects14010042
- Bezerra, F.I. & Mendes, M. (2024) A palaeoecological analysis of the Cretaceous (Aptian) insect fauna of the Crato Formation, Brazil. Palaeogeography, Palaeoclimatology, Palaeoecology, 641, 112134. https://doi.org//10.1016/j.palaeo.2024.112134
- Borror, D.J. & White, R.E. (1970) A field guide to the insects of America north of Mexico. Houghton Mifflin, Boston, 404 pp.
- Brauckmann, C. & Schneider, J. (1996) Ein unter-karbonisches Insekt aus dem Raum Bitterfeld/Delitzsch (Pterygota, Arnsbergium, Deutschland). Neues Jahrbuch für Geologie und Paläontologie-Monatshefte, 1996 (1), 17–30. https://doi.org//10.1127/njgpm/1996/1996/17
- Briggs, D.E.G. (2003) The role of decay and mineralization in the preservation of soft-bodied fossils. Annual Review of Earth and Planetary Sciences, 31, 275–301. https://doi.org//10.1146/annurev.earth.31.100901.144746
- Butler, A.D., Cunningham, J.A., Budd, G.E. & Donoghue, P.C.J. (2015) Experimental taphonomy of Artemia reveals the role of endogenous microbes in mediating decay and fossilization. Proceedings of the Royal Society B: Biological Sciences, 282 (1808), 20150476, 1–10.
- https://doi.org/10.1098/rspb.2015.0476
- Clements, T., Purnell, M. & Gabbott, S. (2019) The Mazon Creek Lagerstätte: A diverse late Paleozoic ecosystem entombed within siderite concretions. Journal of the Geological Society, 176 (1), 1–11. https://doi.org//10.1144/jgs2018-088
- De Jong, R. (2017) Fossil butterflies, calibration points and the molecular clock (Lepidoptera: Papilionoidea). Zootaxa, 4270 (1), 1–63. https://doi.org//10.11646/zootaxa.4270.1.1
- Dlussky, G. & Perfilieva, K. (2014) Superfamily Formicoidea Latreille, 1802. In: Antropov, A.V., Belokobylskij, S.A., Compton, S.G., Dlussky, G.M., Khalaim, A.I., Kolyada, V.A., Kozlov, M.A., Perfilieva, K.S. & Rasnitsyn, A.P. (Eds), The wasps, bees and ants (Insecta: Vespida=Hymenoptera) from the Insect Limestone (Late Eocene) of the Isle of Wight, UK. Earth and Environmental Science Transactions of the Royal Society of Edinburgh, 104 (3-4), 335–446. https://doi.org//10.1017/S1755691014000103
- Duncan, I.J., Titchener, F. & Briggs, D.E.G. (2003) Decay and disarticulation of the cockroach: Implications for preservation of the blattoids of Writhlington (Upper Carboniferous), UK. Palaios, 18 (3), 256–265. https://doi.org//10.1669/0883-1351(2003)018<0256:DADOTC>2.0.CO;2
- Gäb, F., Ballhaus, C., Stinnesbeck, E., Kral, A.G., Janssen, K. & Bierbaum, G. (2020) Experimental taphonomy of fish – role of elevated pressure, salinity and pH. Scientific Reports, 10 (1), 7839. https://doi.org//10.1038/s41598-020-64651-8
- Gibson, B.M., Schiffbauer, J.D., Wallace, A.F. & Darroch, S.A.F. (2023) The role of iron in the formation of Ediacaran ‘death masks’. Geobiology, 21 (4), 421–434. https://doi.org//10.1111/gbi.12551
- Harding, I.C. & Chant, L.S. (2000) Self-sedimented diatom mats as agents of exceptional fossil preservation in the Oligocene Florissant lake beds, Colorado, United States. Geology, 28 (3), 195–198. https://doi.org//10.1130/0091-7613(2000)028<0195:SSDMAA>2.3.CO;2
- Henning, J.T., Smith, D.M., Nufio, C.R. & Meyer, H.W. (2012) Depositional setting and fossil insect preservation: A study of the late Eocene Florissant Formation, Colorado. Palaios, 27 (7), 481–488. https://doi.org//10.2110/palo.2011.p11-084r
- Holden, A.R., Southon, J.R., Will, K., Kirby, M.E., Aalbu, R.L. & Markey, M.J. (2017) A 50,000 year insect record from Rancho La Brea, Southern California: insights into past climate and fossil deposition. Quaternary Science Reviews, 168, 123–136. https://doi.org//10.1016/j.quascirev.2017.05.001
- Iniesto, M., Blanco-Moreno, C., Villalba, A., Buscalioni, Á.D., Guerrero, M.C. & López-Archilla, A.I. (2018) Plant tissue decay in long-term experiments with microbial mats. Geosciences, 8 (11), 387. https://doi.org//10.3390/geosciences8110387
- Iniesto, M., Gutiérrez-Silva, P., Dias, J.J., Carvalho, I.S., Buscalioni, A.D. & López-Archilla, A.I. (2021) Soft tissue histology of insect larvae decayed in laboratory experiments using microbial mats: Taphonomic comparison with Cretaceous fossil insects from the exceptionally preserved biota of Araripe, Brazil. Palaeogeography, Palaeoclimatology, Palaeoecology, 564, 110156. https://doi.org//10.1016/j.palaeo.2020.110156
- Jouault, C., Aase, A. & Nel, A. (2021) Past ecosystems drive the evolution of the early diverged Symphyta (Hymenoptera: Xyelidae) since the earliest Eocene. Fossil Record, 24 (2), 379–393. https://doi.org//10.5194/fr-24-379-2021
- Kim, D.-Y., Lee, M., Nam, G.-S. & Park, T.-Y.S. (2021) The first orthopteran fossils from the Lower Cretaceous (Albian) Jinju Formation of Korea: Ethological implications for elcanids. Cretaceous Research, 125, 104843. https://doi.org//10.1016/j.cretres.2021.104843
- Klompmaker, A.A., Portell, R.W. & Frick, M.G. (2017) Comparative experimental taphonomy of eight marine arthropods indicates distinct differences in preservation potential. Palaeontology, 60 (6), 773–794. https://doi.org//10.1111/pala.12314
- Mähler, B., Janssen, K., Lönartz, M.I., Lagos, M., Geisler, T., Rust, J. & Bierbaum, G. (2023) Time-dependent microbial shifts during crayfish decomposition in freshwater and sediment under different environmental conditions. Scientific Reports, 13 (1), 1539. https://doi.org//10.1038/s41598-023-28713-x
- Martínez-Delclòs, X., Briggs, D.E.G. & Peñalver, E. (2004) Taphonomy of insects in carbonates and amber. Palaeogeography, Palaeoclimatology, Palaeoecology, 203 (1-2), 19–64. https://doi.org//10.1016/S0031-0182(03)00643-6
- McNamara, M.E., Briggs, D.E.G. & Orr, P.J. (2012) The controls on the preservation of structural color in fossil insects. Palaios, 27 (7), 443–454. https://doi.org//10.2110/palo.2012.p12-027r
- Mellmann, J. (2001) The transverse motion of solids in rotating cylinders—forms of motion and transition behavior. Powder Technology, 118 (3), 251–270. https://doi.org//10.1016/S0032-5910(00)00402-2
- Meyer, H.W. (2003) The fossils of Florissant. Smithsonian Institution Press, Washington, D.C., 258 pp.
- Nel, A. & Jouault, C. (2022) The odonatan insects from the Paleocene of Menat, central France. Acta Palaeontologica Polonica, 67 (3), 631–648. https://doi.org//10.4202/app.00960.2021
- Newman, S.A., Daye, M., Fakra, S.C., Marcus, M.A., Pajusalu, M., Pruss, S.B., Smith, E.F. & Bosak, T. (2019) Experimental preservation of muscle tissue in quartz sand and kaolinite. Palaios, 34 (9), 437–451. https://doi.org//10.2110/palo.2019.030
- O’Brien, N.R., Meyer, H.W. & Harding, I.C. (2008) The role of biofilms in fossil preservation, Florissant Formation, Colorado. In: Special paper of the Geological Society of America. Vol. 435. Geological Society of America, Boulder, pp. 19–31.
- O’Brien, N.R., Meyer, H.W., Reilly, K., Ross, A.M. & Maguire, S. (2002) Microbial taphonomic processes in the fossilization of insects and plants in the late Eocene Florissant Formation, Colorado. Rocky Mountain Geology, 37 (1), 1–11. https://doi.org//10.2113/gsrocky.37.1.1
- Penney, D. & Jepson, J.E. (2014) Fossil insects: an introduction to palaeoentomology. Siri Scientific Press, Manchester, UK, 222 pp.
- Petrulevičius, J.F. & Gutierrez, P.R. (2016) New basal Odonatoptera (Insecta) from the lower Carboniferous (Serpukhovian) of Argentina. Arquivos Entomolóxicos, 16, 341–358. https://doi.org//10.5281/zenodo.12765386
- Rajaei, H., Wappler, T., De Jong, R., Wahlberg, N. & Engel, M.S. (2026) Exceptionally preserved Oligocene emperor butterfly from France provides a new calibration point for Apaturinae evolution. Acta Palaeontologica Polonica, 71 (1), 185–191. https://doi.org//10.4202/app.01332.2026
- Rust, J. (2000) Fossil record of mass moth migration. Nature, 405 (6786), 530–531. https://doi.org//10.1038/35014733
- Sansom, R.S., Gabbott, S.E. & Purnell, M.A. (2010) Non-random decay of chordate characters causes bias in fossil interpretation. Nature, 463 (7282), 797–800. https://doi.org//10.1038/nature08745
- Sansom, R.S., Gabbott, S.E. & Purnell, M.A. (2011) Decay of vertebrate characters in hagfish and lamprey (Cyclostomata) and the implications for the vertebrate fossil record. Proceedings of the Royal Society B: Biological Sciences, 278 (1709), 1150–1157. https://doi.org//10.1098/rspb.2010.1641
- Smith, D.M. (2000) Beetle taphonomy in a recent ephemeral lake, southeastern Arizona. Palaios, 15 (2), 152–160. https://doi.org//10.2307/3515501
- Smith, D.M. (2012) Exceptional preservation of insects in lacustrine environments. Palaios, 27 (5), 346–353. https://doi.org//10.2110/palo.2011.p11-107r
- Smith, D.M. & Moe-Hoffman, A.P. (2007) Taphonomy of Diptera in lacustrine environments: A case study from Florissant Fossil Beds, Colorado. Palaios, 22 (6), 623–629. https://doi.org//10.2110/palo.2006.p06-119r
- Smith, D.M., Cook, A. & Nufio, C.R. (2006) How physical characteristics of beetles affect their fossil preservation. Palaios, 21 (3), 305–310. https://doi.org//10.2110/palo.2004.p04-91
- Sohn, J.-C., Labandeira, C.C. & Davis, D.R. (2015) The fossil record and taphonomy of butterflies and moths (Insecta, Lepidoptera): implications for evolutionary diversity and divergence-time estimates. BMC Evolutionary Biology, 15 (1), 12. https://doi.org//10.1186/s12862-015-0290-8
- Sorrentino, J.A., Gibson, B.M., Jouault, C., Archibald, S.B., Piunno, P.A.E. & Laflamme, M. (2026) Controls on the sinking of hymenopterans and orthopterans in water of varying salinity: a taphonomic study. Lethaia, 59 (3), 1–17. https://doi.org//10.18261/let.59.3.1
- Storari, A.P., Osés, G.L., de Almeida-Lima, D.S., Rizzutto, M.A., Bantim, R.A.M., de Lima, F.J., Rodrigues, T. & Sayão, J.M. (2024) Exceptionally well-preserved orthopteran proventriculi from the Cretaceous Crato Formation of Brazil. Journal of South American Earth Sciences, 133, 104737. https://doi.org//10.1016/j.jsames.2023.104737
- Storari, A.P., Salles, F.F., Guedes da Fonseca, J.L., Saraiva, A.Á.F. & Rodrigues, T. (2025) Taphonomy of aquatic insects from the Crato Formation Lagerstätte (Aptian, Lower Cretaceous) under an actualistic look. PLoS ONE, 20 (9), e0331656. https://doi.org//10.1371/journal.pone.0331656
- Ware, J.L., Simonsen, T.J., Rasmussen, J.A. & Archibald, S.B. (2025) Systematics of the extinct suborder Cephalozygoptera (Odonata): a wing-based analysis comparing its genera with those of extant Zygoptera. American Museum Novitates, 4043, 1–20. https://doi.org//10.1206/4043.1
- Wehmann, H.-N., Engels, T. & Lehmann, F.-O. (2022) Flight activity and age cause wing damage in house flies. Journal of Experimental Biology, 225 (1), jeb242872, 1–13. https://doi.org//10.1242/jeb.242872