Abstract
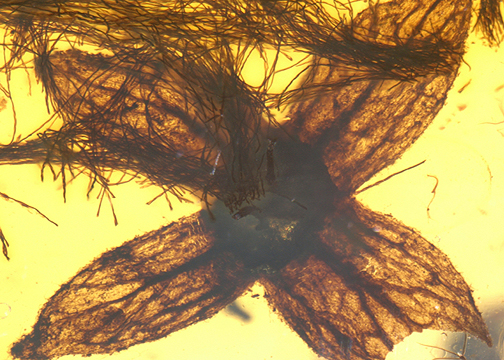
The establishment of a fixed pentamerous floral ground plan was a key innovation in the radiation of core eudicots (Pentapetalae). However, the developmental stability of this trait during the Cretaceous Angiosperm Terrestrial Revolution (ATR) remains poorly understood. Tropidogyne, a dominant flower genus in the mid-Cretaceous Kachin amber, has been previously characterized by a predominantly pentamerous organization and a striking morphological resemblance to the extant family Cunoniaceae (e.g., Ceratopetalum). Here, we report a unique four-tepaled flower assigned to Tropidogyne cf. pentaptera. The reduction from five to four tepals is accompanied by a coordinated decrease in ovary ridges (from ten to eight), suggesting a developmental variation. While this meristic instability is comparable to variation observed in extant Ceratopetalum, the absence of diagnostic cunoniaceous synapomorphies suggests that Tropidogyne likely belongs to an unresolved lineage within rosids. This finding indicates that variability in floral organ number was already present in mid-Cretaceous eudicots. This pattern may reflect developmental plasticity in early eudicot ontogeny and provides new insights into the stabilization of the pentamerous floral ground plan in extant lineages.
References
- Barnes, R.W. & Hill, R.S. (1999) Ceratopetalum fruits from Australian Cainozoic sediments and their significance for petal evolution in the genus. Australian Systematic Botany, 12 (5), 635–645. https://doi.org//10.1071/SB98014
- Benton, M.J., Wilf, P. & Sauquet, H. (2022) The Angiosperm Terrestrial Revolution and the origins of modern biodiversity. New Phytologist, 233 (5), 2017–2035. https://doi.org//10.1111/nph.17822
- Bradford, J.C., Hopkins, H.F. & Barnes, R.W. (2004) Cunoniaceae. In: Kubitzki, K. (Ed.), Flowering plants dicotyledons. The families and genera of vascular plants, vol 6. Springer, Berlin, Heidelberg, pp. 91–111. https://doi.org//10.1007/978-3-662-07257-8_11
- Chambers, K.L., Poinar, Jr., G.O. & Buckley, R. (2010) Tropidogyne, a new genus of Early Cretaceous Eudicots (Angiospermae) from Burmese amber. Novon: A Journal for Botanical Nomenclature, 20 (1), 23–29. https://doi.org//10.3417/2008039
- Condamine, F.L., Silvestro, D., Koppelhus, E.B. & Antonelli, A. (2020) The rise of angiosperms pushed conifers to decline during global cooling. Proceedings of the National Academy of Sciences, 117 (46), 28867–28875. https://doi.org//10.1073/pnas.2005571117
- Cronquist, A. (1981) An integrated system of classification of flowering plants. Columbia University Press, New York, 1262 pp.
- Cruickshank, R.D. & Ko, K. (2003) Geology of an amber locality in the Hukawng Valley, Northern Myanmar. Journal of Asian Earth Sciences, 21 (5), 441–455. https://doi.org//10.1016/S1367-9120(02)00044-5
- Dickison, W.C. (1975) Studies on the floral anatomy of the Cunoniaceae. American Journal of Botany, 62 (5), 433–447. https://doi.org//10.1002/j.1537-2197.1975.tb14068.x
- Ding, W.N., Silvestro, D., Onstein, R.E., Wu, M.X., Zhou, Z.K. & Xing, Y.W. (2025) The stepwise rise of angiosperm-dominated terrestrial ecosystems. Biological Reviews, 100 (5), 2131–2149. https://doi.org//10.1111/brv.70039
- Endress, P.K. (2008) The whole and the parts: relationships between floral architecture and floral organ shape, and their repercussions on the interpretation of fragmentary floral fossils. Annals of the Missouri Botanical Garden, 95 (1), 101–120. https://doi.org//10.3417/2006190
- Endress, P.K. (2011) Evolutionary diversification of the flowers in angiosperms. American Journal of Botany, 98 (3), 370–396. https://doi.org//10.3732/ajb.1000299
- Gandolfo, M.A. & Hermsen, E.J. (2017) Ceratopetalum (Cunoniaceae) fruits of Australasian affinity from the early Eocene Laguna del Hunco flora, Patagonia, Argentina. Annals of Botany, 119 (4), 507–516. https://doi.org//10.1093/aob/mcw283
- Hickey, M. & King, C. (2000) The Cambridge illustrated glossary of botanical terms. Cambridge University Press, Cambridge, 208 pp.
- Hoogland, R.D. (1960) Studies in the Cunoniaceae, I. The genera Ceratopetalum, Gillbeea, Aistopetalum, and Calycomis. Australian Journal of Botany, 8 (3), 318–341. https://doi.org//10.1071/BT9600318
- Huang, D.Y. (Ed.), (2026) Mid-Cretaceous Myanmar amber biota. Shanghai Scientific & Technical Publishers, Shanghai, 121 pp.
- Jud, N.A., Gandolfo, M.A., Iglesias, A. & Wilf, P. (2018) Fossil flowers from the early Palaeocene of Patagonia, Argentina, with affinity to Schizomerieae (Cunoniaceae). Annals of Botany, 121 (3), 431–442. https://doi.org//10.1093/aob/mcx173
- Kitazawa, M.S. & Fujimoto, K. (2014) A developmental basis for stochasticity in floral organ numbers. Frontiers in Plant Science, 5, 545. https://doi.org//10.3389/fpls.2014.00545
- Magallón, S., Crane, P.R. & Herendeen, P.S. (1999) Phylogenetic pattern, diversity, and diversification of eudicots. Annals of the Missouri Botanical Garden, 86 (2), 297–372. https://doi.org//10.2307/2666180
- Magallón-Puebla, S., Herendeen, P.S. & Crane, P.R. (1997) Quadriplatanus georgianus gen. et sp. nov.: staminate and pistillate platanaceous flowers from the Late Cretaceous (Coniacian-Santonian) of Georgia, U.S.A. International Journal of Plant Sciences, 158 (3), 373–394. https://doi.org//10.1086/297448
- Mao, Y.Y., Liang, K., Su, Y.T., Li, J.G., Rao, X., Zhang, H., Xia, F.Y., Fu, Y.Z., Cai, C.Y. & Huang, D.Y. (2018) Various amberground marine animals on Burmese amber with discussions on its age. Palaeoentomology, 1 (1), 91–103. https://doi.org//10.11646/palaeoentomology.1.1.11
- McKim, S.M., Routier-Kierzkowska, A.L., Monniaux, M., Kierzkowski, D., Pieper, B., Smith, R.S., Tsiantis, M. & Hay, A. (2017) Seasonal regulation of petal number. Plant Physiology, 175 (2), 886–903. https://doi.org//10.1104/pp.17.00563
- Monniaux, M, Pieper, B. & Hay, A. (2016) Stochastic variation in Cardamine hirsuta petal number. Annals of Botany, 117 (5), 881–887. https://doi.org//10.1093/aob/mcv131
- Poinar, Jr., G.O. & Chambers, K.L. (2017) Tropidogyne pentaptera, sp. nov., a new mid-Cretaceous fossil angiosperm flower in Burmese amber. Palaeodiversity, 10 (1), 135–140. https://doi.org//10.18476/pale.v10.a10
- Poinar, Jr., G.O. & Chambers, K.L. (2019) Tropidogyne lobodisca sp. nov., a third species of the genus from mid-Cretaceous Myanmar amber. Journal of the Botanical Research Institute of Texas, 13 (2), 461–466. https://doi.org//10.17348/jbrit.v13.i2.798
- Poinar, Jr., G.O., Chambers, K.L. & Vega, F.E. (2021) Tropidogyne euthystyla sp. nov., a new small-flowered addition to the genus from mid-Cretaceous Myanmar amber. Journal of the Botanical Research Institute of Texas, 15 (1), 113–119. https://doi.org//10.17348/jbrit.v15.i1.1053
- Ross, A.J. (2024) Complete checklist of Burmese (Myanmar) amber taxa 2023. Mesozoic, 1 (1), 21–57. https://doi.org//10.11646/mesozoic.1.1.4
- Rozefelds, A.C. & Barnes, R.W. (2002) The systematic and biogeographical relationships of Ceratopetalum (Cunoniaceae) in Australia and New Guinea. International Journal of Plant Science, 163 (4), 651–673. https://doi.org//10.1086/339716
- Schönenberger, J., Friis, E.M., Matthews, M.L. & Endress, P.K. (2001) Cunoniaceae in the Cretaceous of Europe: evidence from fossil flowers. Annals of Botany, 88 (3), 423–437. https://doi.org//10.1006/anbo.2001.1488
- Shi, G.H., Grimaldi, D.A., Harlow, G.E., Wang, J., Wang, J., Yang, M.C., Lei, W.Y., Li, Q.L. & Li, X.H. (2012) Age constraint on Burmese amber based on U-Pb dating of zircons. Cretaceous Research, 37, 155–163. https://doi.org//10.1016/j.cretres.2012.03.014
- Simpson, M.G. (2019) Plant systematics. Academic Press, London, 777 pp.
- Takhtajan, A. (Ed.) (2009) Flowering plants. Springer, Dordrecht, 871 pp. https://doi.org//10.1007/978-1-4020-9609-9
- Wang, D., Dong, X., Zhong, M.C., Jiang, X.D., Cui, W.H., Bendahmane, M. & Hu, J.Y. (2024) Molecular and genetic regulation of petal number variation. Journal of Experimental Botany, 75 (11), 3233–3247. https://doi.org//10.1093/jxb/erae136
- Wang, Y.F. & Manchester, S.R. (2000) Chaneya, a new genus of winged fruit from the Tertiary of North America and eastern Asia. International Journal of Plant Sciences, 161 (1), 167–178. https://doi.org//10.1086/314227
- Yin, Z.W., Cai, C.Y. & Huang, D.Y. (2018) Last major gap in scydmaenine evolution filled (Coleoptera: Staphylinidae). Cretaceous Research, 84, 62–68. https://doi.org//10.1016/j.cretres.2017.10.026