Abstract
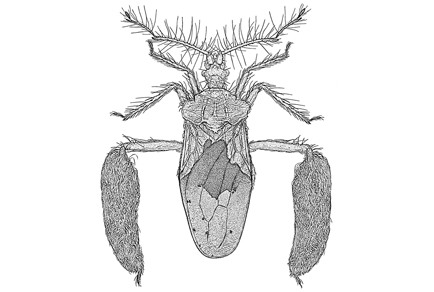
Most adult Holoptilinae in the tribe Holoptilini, including the Australian genus Ptilocnemus Westwood, 1840, have an elaborate glandular structure known as a trichome. This trichome interfaces with its ant prey during predation events. Unlike other genera of Holoptilini though, the nymphs of Ptilocnemus lack any resemblance of a trichome. Here we describe a new species, Ptilocnemus larrakia sp. nov., from the Northern Territory that is contrary to expectations. Although leg colouration, and denser clustering of setae on hind tibia have diagnostic significance, the most striking difference is the unique trichome which includes a new element ‘ptb3’. It is also the first Ptilocnemus species where the nymph is known to possess a trichome during the late instar. This latter observation may have significance for the understanding of the evolutionary history of Ptilocnemus. While the phylogeny of Ptilocnemus and related taxa is yet to be resolved, we speculate that the trichome in the nymphs of Ptilocnemus larrakia sp. nov. may be plesiomorphic, suggesting that this species could be an early diverging species of Ptilocnemus. This finding also suggests that the absence of the gland in the nymphs of other Ptilocnemus species is due to Australian Holoptilini experiencing selective pressures not present in other biogeographical regions.
References
- Andrew, N., L. Rodgerson, and A. York. 2000. Frequent fuel‐reduction burning: the role of logs and associated leaf litter in the conservation of ant biodiversity. Austral Ecology 25: 99–107. https://doi.org/10.1046/j.1442-9993.2000.01015.x
- Amyot, C. J. B. and A. Serville. 1843. Histoire naturelle des insectes. Hémiptères. Librairie encyclopédique de Roret, Paris. 321. https://doi.org/10.5962/bhl.title.8471
- Bardey, D. J., C. Weirauch, R. Hoey-Chamberlain, V. Tyts, N. Tatarnic, S. Arif, and M. W. Bulbert. 2026. The long and short of it: phylogenomics shows loss of myrmecophagous traits in Holoptilinae and necessitates synonymization of feather-legged assassin bug tribes (Hemiptera: Reduviidae). Systematics and Biodiversity 24(1). https://doi.org/10.1080/14772000.2026.2633316
- Bellis, G. A. and A. M. Profke. 2000. Roosting behaviour of the Rainbow Bee-eater Merops ornatus in suburban Darwin. The Northern Territory Naturalist 50: 310.
- Bergstrom, D. M., B. C. Wienecke, J. Van den Hoff, L. Hughes, D. B. Lindenmayer, T. D. Ainsworth, C. M. Baker, L. Bland, D. M. J. S. Bowman, S. T. Brooks, and J. G. Canadell. 2021. Combating ecosystem collapse from the tropics to the Antarctic. Global Change Biology 27: 1692–1703. https://doi.org/10.1111/gcb.15539
- Brennan, I. G., A. R. Lemmon, E. Moriarty Lemmon, C. J. Hoskin, S. C. Donnellan, and J. S. Keogh. 2024. Populating a continent: phylogenomics reveal the timing of Australian frog diversification. Systematic Biology 73: 1–11. https://doi.org/10.1093/sysbio/syad048
- Brennan, K. E., F. J. Christie, and A. York. 2009. Global climate change and litter decomposition: more frequent fire slows decomposition and increases the functional importance of invertebrates. Global Change Biology 15: 2958–2971. https://doi.org/10.1111/j.1365-2486.2009.02011.x
- Buckingham, S., N. Murphy, and H. Gibb. 2019. Effects of fire severity on the composition and functional traits of litter-dwelling macroinvertebrates in a temperate forest. Forest Ecology and Management 434: 279–288. https://doi.org/10.1016/j.foreco.2018.12.030
- Bulbert, M. W., M. E. Herberstein, and G. Cassis. 2014. Assassin bug requires dangerous ant prey to bite first. Current Biology 24(6): 220–R221. https://doi.org/10.1016/j.cub.2014.02.006
- Condamine, F. L., E. F. Toussaint, A. L. Clamens, G. Genson, F. A. Sperling, and G. J. Kergoat. 2015. Deciphering the evolution of birdwing butterflies 150 years after Alfred Russel Wallace. Scientific Reports 5: 11860, 1–11. https://doi.org/10.1038/srep11860
- Crayn, D. M., C. Costion, and M. G. Harrington. 2015. The Sahul–Sunda floristic exchange: dated molecular phylogenies document Cenozoic intercontinental dispersal dynamics. Journal of Biogeography 42: 11–24. https://doi.org/10.1111/jbi.12405
- Cruz, M. G., A. L. Sullivan, J. S. Gould, N. C. Sims, A. J. Bannister, J. J. Hollis, and R. J. Hurley. 2012. Anatomy of a catastrophic wildfire: the Black Saturday Kilmore East fire in Victoria, Australia. Forest Ecology and Management 284: 269–285. https://doi.org/10.1016/j.foreco.2012.02.035
- Dawes‐Gromadzki, T. Z. 2008. Abundance and diversity of termites in a savanna woodland reserve in tropical Australia. Australian Journal of Entomology 47: 307–314. https://doi.org/10.1111/j.1440-6055.2008.00662.x
- Damgaard, J., N. M. Andersen, L. Cheng, and F. A. Sperling. 2000. Phylogeny of sea skaters, Halobates Eschscholtz (Hemiptera, Gerridae), based on mtDNA sequence and morphology. Zoological Journal of the Linnean Society 130(4): 511–526. https://doi.org/10.1111/j.1096-3642.2000.tb02199.x
- de Boer, H. J., K. Steffen, and W. E. Cooper. 2015. Sunda to Sahul dispersals in Trichosanthes (Cucurbitaceae): a dated phylogeny reveals five independent dispersal events to Australasia. Journal of Biogeography 42: 519–531. https://doi.org/10.1111/jbi.12432
- Evans, J. H. 1931. A preliminary revision of the ambush bugs of North America (Hemiptera: Phymatidae). Annals of the Entomological Society of America 24: 711–738. https://doi.org/10.1093/aesa/24.4.711
- Gray, J. E. 1831. Description of a new genus of hemipterous insect, discovered by Major General Hardwicke in India. Zoological Miscellany 1831: 34–35.
- Gibb, H., J. J. Grubb, D. Black, N. Porch, O. Decker, M. McGeoch, D. Deane, and N. Murphy. 2023. Rainforest litter invertebrates decimated by high severity burns during Australia’s gigafires. Austral Ecology 48: 1328–1343. https://doi.org/10.1111/aec.13375
- Holzmeyer, L., F. Hauenschild, and A. N. Muellner‐Riehl. 2023. Sunda–Sahul floristic exchange and pathways into the Southwest Pacific: new insights from wet tropical forest trees. Journal of Biogeography 50: 1257–1270. https://doi.org/10.1111/jbi.14606
- Horváth, G. 1902. Descriptions of new Hemiptera from New South Wales. Természettudományi Füzetek 25: 601–612.
- Jacobson, E. 1911. Biological notes on the hemipteron Ptilocerus ochraceus. Tijdschrift voor Entomologie 54: 175–179.
- Jamali, H., S. J. Livesley, S. P. Grover, T. Z. Dawes, L. B. Hutley, G. D. Cook, and S. K. Arndt. 2011. The importance of termites to the CH₄ balance of a tropical savanna woodland of northern Australia. Ecosystems 14: 698–709. https://doi.org/10.1007/s10021-011-9439-5
- Joyce, E. M. 2021. Evolution of the northern Australian flora: role of the Sunda-Sahul Floristic Exchange. PhD Thesis, James Cook University.
- Köhler, F. and F. Criscione. 2013. Plio‐Pleistocene out‐of‐Australia dispersal in a camaenid land snail. Journal of Biogeography 40: 1971–1982. https://doi.org/10.1111/jbi.12147
- Lawes, M. J., L. A. Woolley, S. Van Holsbeeck, B. P. Murphy, G. E. Burrows, and J. J. Midgley. 2021. Bark functional ecology and its influence on the distribution of Australian half‐butt eucalypts. Austral Ecology 46: 1097–1111. https://doi.org/10.1111/aec.13045
- Lee, G. P. 2003. Mangroves in the Northern Territory. Department of Infrastructure, Planning and Environment, Darwin. Report Number 25/2003D.
- Lepeletier, A. L. M. and J. G. A. Serville, 1825. Histoire naturelle. Entomologie. 280. In G. A. Olivier, ed. Encyclopédie Méthodique, Vol. 10. Mme Veuve Agasse, Paris. 832 pp.
- Maldonado Capriles, J. 1990. Systematic catalogue of the Reduviidae of the world (Insecta: Heteroptera). Caribbean Journal of Science, Special Edition. University of Puerto Rico, Mayagüez. 694 pp.
- Malipatil, M. B. 1983. Revision of Aradellus Westwood: (Heteroptera: Reduviidae: Holoptilinae). The Beagle: Records of the Museums and Art Galleries of the Northern Territory 1(6): 51–56. https://doi.org/10.5962/p.258358
- Malipatil, M. B. 1985. Revision of Australian Holoptilinae (Reduviidae: Heteroptera). Australian Journal of Zoology 33: 283–299. https://doi.org/10.1071/ZO9850283
- Malipatil, M. B. 2018. First record of the genus Ptilocerus in the Australian Region, with the description of two new species (Hemiptera: Heteroptera: Reduviidae). Zootaxa 4410(1): 177–189. https://doi.org/10.11646/zootaxa.4410.1.10
- Marin, J., S. C. Donnellan, S. Blair Hedges, P. Doughty, M. N. Hutchinson, C. Cruaud, and N. Vidal. 2013. Tracing the history and biogeography of the Australian blindsnake radiation. Journal of Biogeography 40: 928–937. https://doi.org/10.1111/jbi.12045
- Masonick, P., A. Michael, S. Frankenberg, W. Rabitsch, and C. Weirauch, 2017. Molecular phylogenetics and biogeography of the ambush bugs (Hemiptera: Reduviidae: Phymatinae). Molecular Phylogenetics and Evolution 114: 225–233. https://doi.org/10.1016/j.ympev.2017.06.010
- Masonick, P. K., A. Knyshov, E. R. Gordon, D. Forero, W. S. Hwang, R. Hoey‐Chamberlain, T. Bush, S. Castillo, M. Hernandez, J. Ramirez, and S. Standring. 2025. A revised classification of the assassin bugs (Hemiptera: Heteroptera: Reduviidae) based on combined analysis of phylogenomic and morphological data. Systematic Entomology 50(1): 102–138. https://doi.org/10.1111/syen.12646
- Mayr, G. L. 1865. Diagnosen neuer Hemipteren II. Verhandlungen der Kaiserlich-Königlichen Zoologisch-Botanischen Gesellschaft in Wie 15: 429–446.
- Miller, N. C. E. 1950. New Reduviidae in the collection of the British Museum (Natural History). IV. Annals and Magazine of Natural History 13: 786–804. https://doi.org/10.1080/00222935008654110
- Mohd-Azlan, J., R. A. Noske, and M. J. Lawes. 2015. The role of habitat heterogeneity in structuring mangrove bird assemblages. Diversity 7: 118–136. https://doi.org/10.3390/d7020118
- Montandon, A. L. 1912. Nouvelles observations sur quelques espèces des genres Ptilocerus Gray et Ptilocnemus Westwood (Holoptilidae). Tijdschrift voor Entomologie 55: 249–254.
- Norman, J. A., L. Christidis, and R. Schodde. 2018. Ecological and evolutionary diversification in the Australo-Papuan scrubwrens (Sericornis) and mouse-warblers (Crateroscelis), with a revision of the subfamily Sericornithinae (Aves: Passeriformes: Acanthizidae). Organisms Diversity & Evolution 18: 241–259. https://doi.org/10.1007/s13127-018-0364-8
- Norman, J. A., F. E. Rheindt, D. L. Rowe, and L. Christidis. 2007. Speciation dynamics in the Australo-Papuan Meliphaga honeyeaters. Molecular Phylogenetics and Evolution 42: 80–91. https://doi.org/10.1016/j.ympev.2006.05.032
- Northern Territory Government. 1999. Charles Darwin National Park Plan of Management. Available from: https://dth.nt.gov.au/parks-and-wildlife/park-management-strategies/management-plans [Last accessed May 25, 2025].
- Oliveros, C. H., D. J. Field, D. T. Ksepka, F. K. Barker, A. Aleixo, M. J. Andersen, P. Alström, B. W. Benz, E. L. Braun, M. J. Braun, and G. A. Bravo. 2019. Earth history and the passerine superradiation. Proceedings of the National Academy of Sciences 116: 7916–7925. https://doi.org/10.1073/pnas.1813206116
- Paradis, E. and K. Schliep. 2019. ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35: 526–528. https://doi.org/10.1093/bioinformatics/bty633
- Redei, D. and J. F. Tsai. 2011. The assassin bug subfamilies Centrocnemidinae and Holoptilinae in Taiwan (Hemiptera: Heteroptera: Reduviidae). Acta Entomologica Musei Nationalis Pragae 51: 215–232.
- Rodrigo, A., F. Sardà-Palomera, J. Bosch, and J. Retana. 2008. Changes of dominant ground beetles in black pine forests with fire severity and successional age. Ecoscience 15: 442–452. https://doi.org/10.2980/15-4-3117
- Rowe, K. C., M. L. Reno, D. M. Richmond, R. M. Adkins, and S. J. Steppan. 2008. Pliocene colonization and adaptive radiations in Australia and New Guinea (Sahul): multilocus systematics of the old endemic rodents (Muroidea: Murinae). Molecular Phylogenetics and Evolution 47: 84–101. https://doi.org/10.1016/j.ympev.2008.01.001
- Saunders, M. E., P. S. Barton, J. R. Bickerstaff, L. Frost, T. Latty, B. D. Lessard, E. C. Lowe, J. Rodriguez, T. E. White, and K. D. Umbers. 2021. Limited understanding of bushfire impacts on Australian invertebrates. Insect Conservation and Diversity 14: 285–293. https://doi.org/10.1111/icad.12493
- Savage, J. M. 1973. The geographic distribution of frogs: patterns and predictions, pp. 351–445. In Vial, J. L., ed. Evolutionary biology of the anurans: contemporary research on major problems. University of Missouri Press, Columbia, Missouri. 470 pp.
- Schliep, K. P. 2011. Phangorn: phylogenetic analysis in R. Bioinformatics 27: 592–593. https://doi.org/10.1093/bioinformatics/btq706
- Schliep, K., A. A. Potts, D. A. Morrison, and G. W. Grimm. 2017. Intertwining phylogenetic trees and networks. Methods in Ecology and Evolution 8: 1212–1220. https://doi.org/10.1111/2041-210X.12760
- Stål, C. 1874. Enumeratio Hemipterorum: Bidrag till en förteckning öfver alla hittills kända Hemiptera, jemte systematiska meddelanden. P. A. Norstedt, Stockholm. 187 pp.
- Swinstead, C., P. Nevill, and P. W. Bateman. 2025. Can epigeic invertebrates escape fire? Pacific Conservation Biology 31: PC24051, 1–7. https://doi.org/10.1071/PC24051
- Toussaint, E. F., R. Tänzler, C. Rahmadi, M. Balke, and A. Riedel. 2015. Biogeography of Australasian flightless weevils (Curculionidae, Celeuthetini) suggests permeability of Lydekker’s and Wallace’s Lines. Zoologica Scripta 44: 632–644. https://doi.org/10.1111/zsc.12127
- Tsang, S. M., S. Wiantoro, M. J. Veluz, N. Sugita, Y. L. Nguyen, N. B. Simmons, and D. J. Lohman. 2020. Dispersal out of Wallacea spurs diversification of Pteropus flying foxes, the world’s largest bats (Mammalia: Chiroptera). Journal of Biogeography 47: 527–537. https://doi.org/10.1111/jbi.13750
- Walker, F. 1873. Catalogue of the specimens of heteropterous-Hemiptera in the collection of the British Museum. British Museum. London. 7:1–213.
- Weirauch, C. and G. Cassis. 2006. Attracting ants: the trichome and novel glandular areas on the sternum of Ptilocnemus lemur (Heteroptera: Reduviidae: Holoptilinae). Journal of the New York Entomological Society 114: 28–37. https://doi.org/10.1664/0028-7199(2006)114[28:AATTAN]2.0.CO;2
- Weirauch, C., M. Bulbert, and G. Cassis. 2010. Comparative trichome morphology in feather-legged assassin bugs (Insecta: Heteroptera: Reduviidae: Holoptilinae). Zoologischer Anzeiger 248: 237–253. https://doi.org/10.1016/j.jcz.2009.03.004
- Westwood, J. O. 1840. Memoir on the genus Holoptilus. Transactions of the Entomological Society 2: 248–253.
- Wightman, G. M. 2006. Mangroves of the Northern Territory, Australia: identification and traditional use. Northern Territory Botanical Bulletin 31: 1–190.
- Wikars, L. O. and J. Schimmel. 2001. Immediate effects of fire-severity on soil invertebrates in cut and uncut pine forests. Forest Ecology and Management 141: 189–200. https://doi.org/10.1016/S0378-1127(00)00328-5